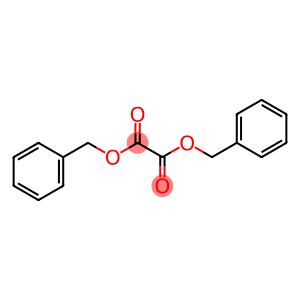
dibenzyl ethanedioate
Dibenzyl oxylate
CAS: 7579-36-4
Molecular Formula: C16H14O4
dibenzyl ethanedioate - Names and Identifiers
| Name | Dibenzyl oxylate |
| Synonyms | Benzyl oxalate Dibenzyl oxylate DIBENZYL OXALATE dibenzyl ethanedioate oxalic acid dibenzyl ester Ethanedioic acid bis(phenylmethyl) ester Ethanedioic acid,1,2-bis(phenylmethyl) ester |
| CAS | 7579-36-4 |
| EINECS | 411-720-3 |
| InChI | InChI=1/C16H14O4/c17-15(19-11-13-7-3-1-4-8-13)16(18)20-12-14-9-5-2-6-10-14/h1-10H,11-12H2 |
dibenzyl ethanedioate - Physico-chemical Properties
| Molecular Formula | C16H14O4 |
| Molar Mass | 270.28 |
| Density | 1.212 |
| Melting Point | 80-82 °C (lit.) |
| Boling Point | 235 °C/14 mmHg (lit.) |
| Flash Point | 171°C |
| Vapor Presure | 3.01E-06mmHg at 25°C |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.5447 (estimate) |
dibenzyl ethanedioate - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| HS Code | 29171100 |
dibenzyl ethanedioate - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Introduction | dibenzyl oxalate is usually white flaky crystals, insoluble in water, with small coloring and low melting point, chemical stability, is an important organic synthesis intermediates. |
| Application | dibenzyl oxalate is an important intermediate in organic synthesis due to its low melting point and stable chemical properties, can be used for the synthesis of sedative benzoyl phenobarbital. |
| Use | dibenzyl oxalate is a sensitizer; It can be used to prepare composite color-changing temperature fibers. |
| preparation method | in a ML three-necked flask equipped with a thermometer, a water trap and a reflux condenser, add the experimental amount of oxalic acid, benzyl alcohol, the water-carrying agent was heated and stirred until the oxalic acid was completely dissolved; Then a certain amount of catalyst was added, and heated and refluxed to generate anhydrous beads in the water separator, indicating that the reaction was completed. The reactor was then changed to a distillation apparatus, most of the water-carrying agent was distilled off, and the remaining reaction solution was poured into 100g of crushed ice while hot, I .e., a large amount of white precipitate was generated. The mixture was allowed to stand until the precipitate was complete. The filter cake was washed twice with distilled water and dried. The filter cake was taken out and recrystallized with 50ml of ethanol, I .e., white flaky crystals were precipitated, suction filtered, dried, weighed, and the yield of oxalic acid dibenzyl Ester was calculated. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Dibenzyl Oxalate Visit Supplier Webpage Request for quotationCAS: 7579-36-4
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Dibenzyl oxalate Visit Supplier Webpage Request for quotationCAS: 7579-36-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Dibenzyl oxalate Request for quotationCAS: 7579-36-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Dibenzyl oxylate Request for quotation
CAS: 7579-36-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 7579-36-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Dibenzyl Oxalate Visit Supplier Webpage Request for quotation
CAS: 7579-36-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7579-36-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Dibenzyl Oxalate Visit Supplier Webpage Request for quotationCAS: 7579-36-4
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Dibenzyl oxalate Visit Supplier Webpage Request for quotationCAS: 7579-36-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Dibenzyl oxalate Request for quotationCAS: 7579-36-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Dibenzyl oxylate Request for quotation
CAS: 7579-36-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 7579-36-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Dibenzyl Oxalate Visit Supplier Webpage Request for quotation
CAS: 7579-36-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7579-36-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




